A short History of Onchocerciasis
by Guido Kluxen
Discovery of the microfilariae
In 1874, John O’Neill, a British naval surgeon attached to HMS Decoy at Cape Coast Castle in the Gold Coast (Ghana) became intrigued by an irritating and intractable skin disease somewhat resembling scabies which afflicted many people living in parts of the West Coast of Africa. He determined to look for the cause of this peculiar condition, which was known locally as “craw-craw”, by studying a number of patients in Addah Fort Hospital under the care of Dr. Thompson of the Glover Expedition. The condition was characterized by papules, vesicles and pustules. O’Neill examined the contents of pustules and vesicles under a microscope but found nothing other than leucocytes. When he turned his attention to papules, success attended his efforts for he found an organism which he had no doubt, was the cause of the complaint. He reported his observations in 1875.
When specimen were examined in a drop of water under a microscope, microfilariae that were easily detectable by virtue of their violent contortions were often seen: “At the head, or blunted extremity, two small dots are noticed, but their nature could not be determined” (Fig. 1) (O’Neill 1875).
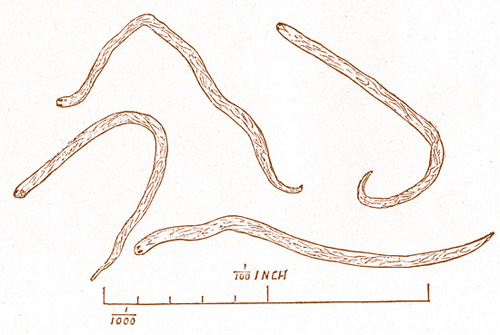 Fig. 1: Unsheathed microfilariae from “craw-craw”
Fig. 1: Unsheathed microfilariae from “craw-craw”
of O’Neill at the Gold Coast, Ghana (Original from The Lancet 1875).
Discovery of the adult worm

Fig. 2: Rudolf Leuckart (1822-1898), zoologist and helminthologist in Leipzig, Germany, discovered Onchocerca volvulus
Onchocerca volvulus:
"The twisting hook-tail"
Leuckart had also sent Manson a histological section containing a fragment of the uterus of one of the worms, Manson remarked: “… I did not see represented in the embryo of Filaria volvulus the sheath. Professor Leuckart makes the same remark.”
The name Onchocerca was derived from a combination of the Greek word ONCHOS meaning “hook” and KERKOS; CERCOS meaning “tail” (Railliet and Henry 1910).
Symptomentrias according to Robles
The first persons to mention ocular onchocerciasis were Rodolfo Robles and Pacheco-Luna in Guatemala in 1915/1916. Ophthalmic symptoms and signs were marked in the Triassic, also known as ‘Morbus Robles’: 1. Filarial worm infection of an adult Onchocerca in America, 2. Erisipela de la costa, red skin inflammation of the coast, a skin disease located on the face, 3. Conjunctivitis and iritis of the anterior segment of the eye (Pacheco Luna 1918).
In 1915, Robles was consulted by a women concerning recurrent erysipelas of the face which was accompanied by fever, a burning sensation, pruritus and poor vision (Fig. 4), but he did not know the cause. Afterwards he saw a boy with the same features: Oedema of the eyelids, the forehead, and the superior lip. On his forehead was a tumour the size of a cherry. It was this lump that Robles excised and first discovered the adult Onchocerca in the Americas and later remarked: “I understood then that the ‘erysipelas’ lesions surely were due to the presence of this parasite” (Robles 1917).
In the coffee-producing districts, having altitudes between 2,500 to 5,000 feet, over 95 percent of the population in an area of volcanoes are Indians who are especially exposed to the bites of Simuliidae and an infection with onchocerciasis.
Fig. 3: Rodolfo Robles discovered onchocerciasis
of the Americas in Guatemala

Fig. 4: In 1915, Rodolfo Robles was consulted by this women
with an erysipelas of the face as the reason was still unknown.
Brumpt gave a detailed description of the parasite (Brumpt 1919), and after comparing it with the specimens collected by him in the Congo, decided that it was a different species which he named O. caecutiens to indicate that the parasite caused blindness (from the Latin “caecus” = blind). But the two were morphologically indistinguishable and there was no consistent difference existing between them.
Fig. 5: Émile Brumpt (1877-1951)
It was the itching and consequent scratching which led to the rediscovery by Montpellier and Lacroix (1920) of microfilariae in the integument which were the same as O’Neill’s parasites. But Brumpt could not believe it (Brumpt 1920). In the middle of the 1920’s, it was becoming clear that the unsheathed microfilariae obtained from O. volvulus adults were indeed the same microfilariae seen in the skin. Finally, despite the reservations of some sceptics such as Brumpt, it seemed certain in retrospect that the filariae seen in the skin 50 years earlier by O’Neill were indeed O. volvulus microfilariae.
Montpellier and Lacroix constantly found microfilariae in the dermal layer of the skin examining members of native troops in Africa suffering with a form of itch or “craw-craw”. They concluded that the “craw-craw” described by O’Neill is a dermal manifestation of onchocerciasis termed “gâle filarienne” (Fig. 6).
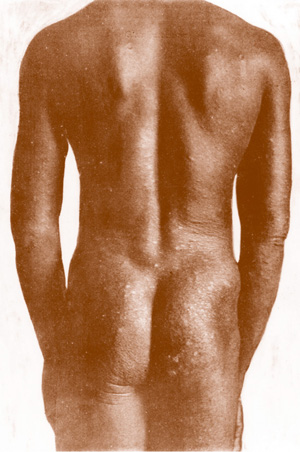
Fig. 6: Gâle filarienne (Original from Montpellier and Lacroix 1920)

Fig. 7: Donald Breadalbane Blacklock (1879-1955), born in Scotland .
Simulium damnosum Theobald:
Mode of transmission of onchocerciasis
In 1923, the Briton, Donald Breadalbane Blacklock, began to investigate the mode of transmission of onchocerciasis in Sierra Leone. Since the microfiariae were not in the blood but in the skin, he postulated that any arthropode capable of transmitting the worms must be able to damage the skin and dislodge the larvae in its efforts to reach blood. Accordingly, he first looked at the Congo floor maggot, Auchmeromyia luteola, which was common in the houses, but no signs of the parasites were found. In 1923/24, he observed that the blackfly prophetically named Simulium damnosum by Theobald in 1903 (Theobald 1903) was biting viciously and in great numbers near the streams supplying several of the villages in an endemic area of onchocerciasis. Furthermore, he noticed that the insect was slow in drawing blood, which reinforced his idea that it must be inflicting severe damage. He therefore caught 100 specimens and examined them for larvae, but finding nothing, abandoned the search temporarily. In 1925, he resumed this operation at another village, this time with success. 780 flies were captured while biting randomly selected boys and 2.6 % of the insects contained larvae morphologically identical with O.volvulus microfilariae. Thereupon, he submitted two men who were known to have O.volvulus microfilariae in their skin, but were not infected with any other filarial parasite, to the flies and found that 17 % of Simuliidae contained microfilariae. When flies were permitted to bite only on a 4 inch band around the patients’ body, this area including nodules near the trochanters, 80 % of the insects became infected. The development of the L3-larvae in the fly is demonstrated in Blacklock’s original figure (Fig. 8). Only the female was found biting, and the biting habit is diurnal (Blacklock 1926/1927).
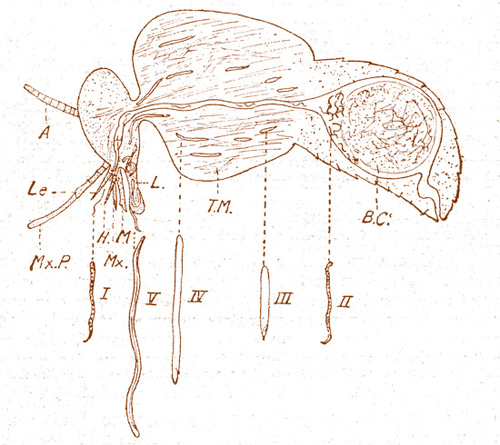
Fig. 8: Diagram to illustrate the development of O. volvulus larvae in the tissues of Simulium damnosum (Blacklock 1927). I = Larva from infected skin, II = larva in mid-gut, III = larva in thoracic muscles (early stage), IV = Larva in thoracic muscles (late stage), V = Larva in head and labium, A = Antenna, B.C. = Blood coagulum, T.M. = Thoracic muscles, L = Labium, L.e. = Labrum-epipharynx, H. = Hypopharynx, M. = Mandible, Mx. = Maxilla, Mx.P. = Maxillary palp. The infective stage larvae (V) are now termed L3 – larvae, and stage-I-larvae are now termed microfilariae.

Fig. 9: Simulium damnosum Theobald (Photo: Hj. Trojan)
The observations of Hissette radically changed the way
of looking at onchocerciasis
The early investigators of onchocerciasis from 1874-1930 in Africa made no mention at all of a concomitant severe eye disease. Publications of the observations in Central America by Rodolfo Robles and Émile Brumpt in 1917/1919 prompted some specialists in tropical medicine to look for eye disease associated with African onchocerciasis. However, there did not seem to be any eye disease leading to blindness (Fülleborn 1924). Only Ouzilleau and his colleagues recorded in 1921 that one of the 16 infected persons of 27 inhabitants they had found in a village near Brazzaville had keratitis. Blacklock (1927) reported that he could find no evidence of eye disease in patients with onchocerciasis in Sierra Leone.
It was not until 1930/1931 that Jean Hissette reported that 20% of patients with onchocerciasis were blind in an onchocerciasis focus on the Sankuru River in the Belgian Congo, and that 50% of the villagers suffered from eye troubles. Two years later, he found a second focus with the same pathology on the Uéle River. Hissette described the pathomechanism of this blindness in a long paper in 1932 and in addition to a description of anterior eye disease, he drew attention to an association with chororetinitis. Two years later the Harvard African Expedition to the Belgian Congo under Richard Pearson Strong with five American colleagues was organized at Belgium’s expense. His members had to check Dr. Jean Hissette’s reports because there were doubts about his findings. Confronted by this commission in the form of the Harvard African Expedition, Hissette traveled once more to the Sankuru with the Americans as the seventh member of the expedition and showed them "his" river blindness patients. The Americans finally confirmed all the observations on river blindness caused by onchocerciasis in the American Journal of Tropical Medicine (Strong 1938) that had already been communicated by Hissette (1932).

Fig. 10: Riverblind people from the Sankuru Region 1930
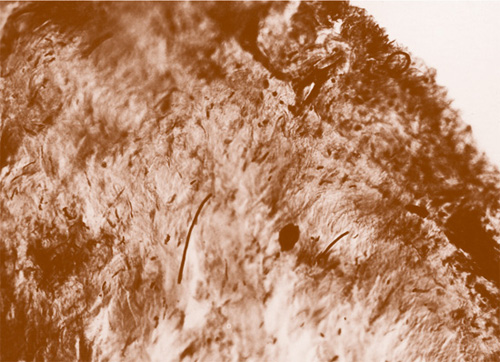
Fig. 11: Microfilariae of Onchocerca volvulus in the Chorioidea of an enucleated eye from the Sankuru River 1931 (Original histology by Jean Hissette, microfilariae are black and stretched)
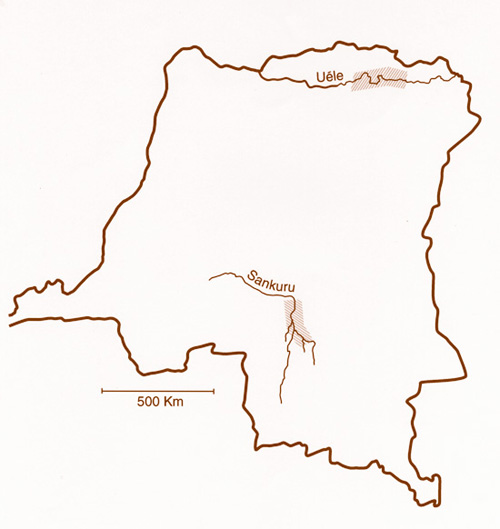
Fig. 12: The Sankuru River Region and Uéle River Region of the Belgian Congo
Hissette’s findings prompted renewed efforts to find ocular complications of onchocerciasis in other parts of Africa, now known as river blindness. In 1944, Harold Ridley found that slightly more than one third of patients with onchocerciasis in a region of the Gold Coast (Ghana) had evidence of either anterior or posterior disease of the eye, with nearly half of them being blind or nearly blind. Ridley’s monograph Ocular onchocerciasis (1945) was of considerable success in river blindness research.
The numerical distribution of microfilariae of Onchocerca volvulus in the skin of man follows a clear pattern which is related to the distribution and severity of the skin and eye lesions. For the eye changes and blindness this is especially the region of the head and neck (Kershaw et al. 1954).
Choyce (1958) confirmed that the scared fundus, hitherto thought to be due to onchocerciasis, was identical with the chorioidal sclerosis described by Sorsby (1939). This could be the fact in Bonjongo, Cameroons where he found such changes indistinguishable for him. Sorsby’s chorioidal sclerosis is a genetic disease transmitted in a dominant fashion. No evidence was found of vitamin A deficiency as a cause of chorioretinitis in onchocerciasis (Woodruff et al. 1963).
Onchocerca volvulus has not been transmitted experimentally to humans by infected flies, but epidemiological evidence suggests that the prepatent period is between 3 to 18 months. The adult worms may live for 15 years and are capable of producing microfilariae for up to ten years (Roberts et al. 1967), while microfilariae may persist for from 6 months to 3 years (Duke 1968).
On October 21, 1987, Merck officials (MSD Merck Sharp & Dohme) said that they would be donating their drug Ivermectin to affected areas “for as long as it might be needed”. Diethylcarbamazine and suramin caused severe side effects in so many individuals. Ivermectin can be given as a tablet on a once a year basis and has minimal side effects. The drug is distributed by Merck, in close collaboration with WHO, an independent committee of experts in tropical medicine, The World Bank, The Carter Centre in Atlanta, Georgia/USA, and about a dozen NGDO’s (non-governmental developing organizations) in several different countries.
Most of the evidence supporting a role for Wolbachia in the pathogenesis of filarial diseases stems from adverse reactions in infected individuals. For example, systemic treatment of onchocerciasis patients with diethylcarbamazine (DEC) causes rapid death of the microfilariae in the skin and eyes, resulting often severe post treatment side effect the so called “Mazzotti reaction”. The severity of the “Mazzotti reaction” is dependent on the number of microfilariae containing Wolbachia in the skin and eyes (Hoerauf and Pfarr 2007). And this was also the fact when Hissette induced the erysipelas in a little 12 year old boy at the Uéle in 1933 (Hissette 1933, Kluxen 2011) by needling his head nodules and killing the worms in it. Reactions include fever, headache, dizziness, myalgia, arthralgia, tachycardia, ciliary injection, severe pruritus, enlargement of lymph nodes and erysipelas of the coast.
While alive, the microfilariae appear to cause little or no inflammation, even being in the anterior chamber. However, when they die, either by natural attrition or after chemotherapy, the host response to degenerating worms can result in ocular inflammation (keratitis, uveitis, chorioretinitis, optic neuritis) that causes progressive loss of vision. Blindness therefore tends to occur in adulthood after many years of infection.